Research Grade” vs Patient-Intended Peptides: What “Physician Use Only” Really Means (and Why Supervised Care Matters)
Written by
TRTBOSS Editorial Team

Introduction to Peptides
Peptides are short chains of amino acids that can act as signaling molecules in the body. In clinical medicine, some peptide-based medications are regulated prescription drugs, while other peptide products exist in a gray market, often advertised online as “research grade,” “not for human consumption,” or “physician use only.”
Those labels can sound reassuring. But they often signal the opposite: the product may not be legally marketed for patient administration, may not meet patient-intended quality standards, and may not come with the clinical safeguards that help reduce avoidable risk.
This article explains the practical, legal, and safety differences between (1) buying “research grade / physician use only” peptides on the internet and (2) receiving peptide-based medication within a TRTBOSS care program, where a licensed clinician evaluates eligibility, prescribes when appropriate, and a licensed pharmacy dispenses medication intended for patient use.
What “peptide therapy” usually means in real life
In consumer wellness culture, “peptide therapy” is used as an umbrella term for a wide range of products. Some legitimate prescription medications, some compounded preparations, and some unapproved substances sold as “research chemicals.”
From a clinician’s perspective, the key question is not whether something is called a “peptide.” The key question is:
Is this a medication intended for patient use under a valid prescription, produced and dispensed within a regulated healthcare and pharmacy framework?
If not, the risk profile changes dramatically, especially for products that are injected or otherwise bypass normal protective barriers (skin, GI tract).
Why the “research grade” market exists
Many “research use only” sellers position themselves as laboratory suppliers. They may use disclaimers like:
-
“For research purposes only”
-
“Not for human consumption”
-
“Physician use only”
But the FDA has repeatedly warned that some websites selling these products to U.S. consumers are marketing unapproved and misbranded drugs and that selling them for consumer use can violate the Federal Food, Drug, and Cosmetic Act. FDA
What the risk signals look like
1) “Not for human consumption” is not a formality, it’s a warning label
When a product is truly produced for laboratory experiments, it may not be manufactured under patient-intended standards for:
-
Sterility assurance (critical for injections)
-
Endotoxin control
-
Identity/purity verification to clinical thresholds
-
Stability and beyond-use dating under healthcare standards
-
Traceability, recalls, adverse-event handling, and accountability
These are not theoretical issues. Public health guidance repeatedly emphasizes how lapses in sterile technique and injection safety can cause serious harm. CDC
2) Online, unregulated drug sales have a measurable quality problem
In the broader “no-prescription online” marketplace, published research has documented quality and safety risks, including mismatch between labeled and measured contents and counterfeit characteristics in products purchased through illegal channels.PubMed
Even if a seller provides a “COA” (certificate of analysis), that document may be incomplete, non-verifiable, not lot-specific, or based on limited testing. A COA is not the same thing as a medication supply chain that is designed for patients.
3) Counterfeit and misbranded medicines are a recognized public safety issue
The FDA warns that counterfeit medicines may contain the wrong ingredients, too much/too little active ingredient, or harmful ingredients. FDA
This risk is amplified when products are purchased outside normal prescription channels.
The Core Difference: Product Category vs Care System
It helps to separate two things:
The substance (a peptide)
The system it travels through (evaluation → prescribing → dispensing → monitoring)
A peptide bought as a “research chemical” is not simply “the same thing, cheaper.” It’s in a different regulatory category. Typically outside the patient-intended safety and accountability framework.
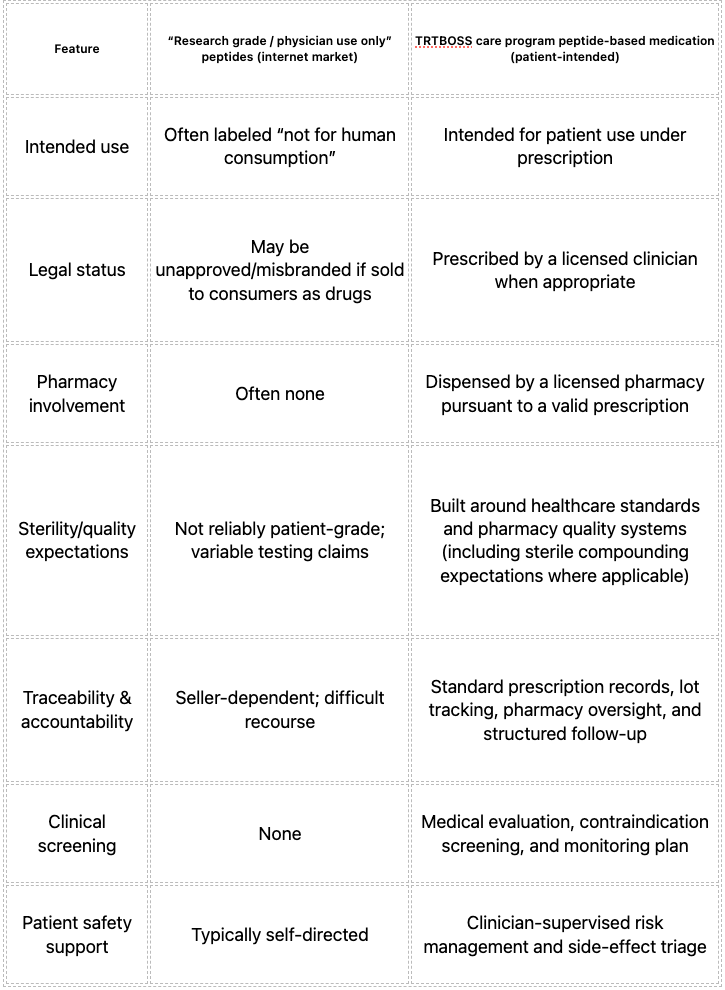
A side-by-side comparison
Safety & Risks
Why peptides are uniquely “high-risk” when purchased outside medical care
A major risk driver is route of administration. Many peptides in the gray market are promoted for injection. With injectable products, the safety bar is higher because problems can show up fast and severely:
-
contamination (microbial or endotoxin-related)
-
incorrect concentration or dosing confusion
-
inappropriate technique, unsafe injection practices
-
unexpected interactions with existing conditions or medications
CDC guidance underscores that safe injection practices are foundational to preventing healthcare-associated infections and other harms. CDC
“Physician use only” can be misleading
“Physician use only” is sometimes used as marketing language. In legitimate healthcare, “physician use” typically means the product is handled within licensed clinical or pharmacy settings, with proper storage, traceability, and professional oversight.
A website banner is not a substitute for:
-
a real prescription relationship
-
pharmacy licensing
-
established quality systems
-
patient counseling and monitoring
Compounding realities (important nuance)
Some peptide-based medications may be compounded in certain contexts. Compounding is regulated, but it is not the same as mass manufacturing of FDA-approved drugs.
FDA resources explain compounding policies and the 503A/503B framework (patient-specific pharmacy compounding vs outsourcing facilities). FDA
In sterile compounding discussions, preventing contamination, endotoxins, and potency variation is a central theme. ASHP
The key point for patients: compounded medications, when used, should still come from a licensed pharmacy and be tied to a clinician’s prescription and follow-up plan, not from a consumer “research chemical” supply chain.
Telehealth Process: How TRTBOSS Approaches Peptide-Based Care
TRTBOSS is structured to keep peptide-based medication (when clinically appropriate) inside a medical care framework:
1) Medical intake and eligibility screening
A clinician-led program starts with medical history, goals, and risk factors. The aim is to identify situations where peptide-based medication may be inappropriate or higher risk.
2) Clinical decision-making (non-prescriptive, individualized)
If a licensed clinician determines a prescription is appropriate, it’s based on individual factors, not influencer protocols or forum dosing.
3) Licensed pharmacy dispensing (patient-intended medication)
Medication intended for patient use is dispensed through a licensed pharmacy pursuant to a valid prescription, creating traceability and accountability that “research-grade” sellers typically do not provide.
4) Ongoing monitoring and safety check-ins
A medical program includes follow-up: side-effect assessment, response monitoring, and adjustments when clinically warranted.
If you’re considering peptide-based therapy, the safest first step is a clinician-guided evaluation, not a checkout cart. Find our clinician approved prescription peptides below, or at www.trtboss.com
Who should be especially cautious
This isn’t a checklist for self-diagnosis. It’s a safety lens.
Extra caution is warranted if you have any of the following:
-
history of severe allergic reactions
-
autoimmune or complex inflammatory conditions
-
endocrine disorders
-
kidney or liver disease
-
cardiovascular disease risk
-
use of multiple prescription medications (interaction potential)
-
needle anxiety or low confidence in injection technique
-
prior injection site infections or complications
Bottom line: A “research peptide” purchase removes the exact safeguards that matter most for higher-risk individuals.
Practical Red Flags When Evaluating Any Peptide Product
If you’re trying to decide whether something is patient-appropriate, these are meaningful warning signs:
-
“Not for human consumption” anywhere on the label or listing
-
No prescription required
-
No licensed pharmacy dispensing
-
No clinician relationship or follow-up plan
-
Vague sourcing (“lab tested” with no lab identity, no lot, no methods)
-
Unverifiable COAs or COAs that don’t match the lot you received
-
No cold-chain clarity for temperature-sensitive products
-
Telegram/DM-based “support” instead of medical oversight
FDA enforcement actions against online sellers illustrate that these patterns can overlap with illegal sale of unapproved/misbranded drugs. FDA
Frequently Asked Questions
What does “research use only” mean for peptides?
It generally indicates the product is marketed for laboratory research, not for administration to patients. Products labeled this way may not meet patient-intended manufacturing, sterility, labeling, and accountability standards.
2) Is “physician use only” the same as prescription-only?
Not necessarily. “Physician use only” can be marketing language. Prescription-only medication should be tied to a licensed clinician’s prescription and dispensed by a licensed pharmacy within regulated channels.
3) Are “research grade” peptides tested and safe?
Some may be tested for limited parameters, but testing claims vary widely and may be difficult to verify. A COA is not equivalent to a patient-intended medication supply chain or ongoing medical monitoring.
4) Why is injection risk higher with non-patient-intended products?
Injectables require reliable sterility and safe injection practices. Public health guidance emphasizes that failures in injection safety and aseptic technique can cause serious harm.
5) What makes a TRTBOSS peptide medication “intended for patient use”?
When clinically appropriate, a licensed clinician evaluates you and writes a prescription, and a licensed pharmacy dispenses medication pursuant to that prescription—creating traceability, counseling, and follow-up pathways.
6) Does clinician supervision matter if I “know what I’m doing”?
Clinician supervision adds safety layers that self-directed use can’t replicate: contraindication screening, interaction review, adverse-effect triage, and structured monitoring.
7) How can I reduce risk if I’m exploring peptide-based therapy?
Choose a clinician-guided program, verify that medication is dispensed by a licensed pharmacy with a prescription, and avoid products labeled “not for human consumption.”
Medical Disclaimer
This content is for educational purposes only and does not constitute medical advice, diagnosis, or treatment. Decisions about medication and weight management should be made with a licensed healthcare professional who can evaluate your individual health history, risks, and goals. If you have severe symptoms (such as persistent vomiting, severe abdominal pain, chest pain, or signs of dehydration), seek urgent or emergency care.
Sources
FDA – Counterfeit Medicine: https://www.fda.gov/drugs/buying-using-medicine-safely/counterfeit-medicine
FDA – Buying Medicines Online: https://www.fda.gov/drugs/buying-using-medicine-safely/buying-medicines-online
FDA – BeSafeRx: Know Your Online Pharmacy: https://www.fda.gov/drugs/buying-using-medicine-safely/besaf-erx-know-your-online-pharmacy
FDA – Human Drug Compounding: Policies and Rules: https://www.fda.gov/drugs/human-drug-compounding/human-drug-compounding-policies-and-rules
CDC – Injection Safety: Clinical Safety: https://www.cdc.gov/injection-safety/hcp/clinical-safety/index.html
CDC – Injection Safety: Clinical Guidance: https://www.cdc.gov/injection-safety/hcp/clinical-guidance/index.html
NABP – Safe.Pharmacy (legitimate online pharmacy guidance): https://safe.pharmacy/
World Health Organization – Substandard and Falsified Medical Products: https://www.who.int/news-room/fact-sheets/detail/substandard-and-falsified-medical-products
ASHP – Compounding (practice resources): https://www.ashp.org/Pharmacy-Practice/Compounding
Prescription Human Grade Peptides
Written by TRTBOSS Editorial Teaam
Related Articles
TRTBOSS Editorial Team in Sexual Health
Research Grade” vs Patient-Intended Peptides: What “Physician Use Only” Really Means (and Why Supervised Care Matters)
Table of contents Introduction to Peptides What “peptide therapy” usually means in real life Why the “research grade” market exists What the risk signals look like 1) “Not for human consumption” is not a formality, it’s a warning label 2) Online, unregulated drug sales have a measurable quality problem 3) Counterfeit and misbranded medicines are […]
TRTBOSS Editorial Team in Blood Panel
Why Insurance Often Doesn’t Cover GLP-1 Medications for Weight Loss (and What TRTBOSS Does Instead)
If you’ve looked into GLP-1 receptor agonists (and related “incretin-based” medicines) for chronic weight management, you may have run into a frustrating reality: many insurance plans won’t cover them for weight loss, even when a clinician agrees the medication could be appropriate. This is not simply a paperwork problem. Coverage decisions for weight management medications […]
Felipe Gougeon, RN, MPH, BScRS-PT in Longevity
Why You Should Avoid “Research Use Only” Peptides
As a registered nurse, and a bachelor in rehabilitation sciences – physical therapy, I’ll say this plainly: when a vial says “for research use only” (RUO) or “not intended for human consumption,” that’s not marketing fluff, it’s a safety boundary. Those labels usually mean the product is being sold as a lab reagent, not as […]
Start Your Health Assessment Today
Personalized telehealth treatments tailored for you.